This website uses cookies so that we can provide you with the best user experience possible. Cookie information is stored in your browser and performs functions such as recognising you when you return to our website and helping our team to understand which sections of the website you find most interesting and useful.
The Digital Cervical Cell Analysis Solution, miLab™ CER

An AI-powered digital cytology solution
that automates the conventional Pap smear workflow
miLab™ CER is attracting attention as an innovative digital cytology platform that significantly reduces the complexity of conventional cervical cytology procedures and the dependency on expert personnel, while enabling high-quality automated diagnostics. In the WHO–UNITAID Technology Landscape Report (2024)*, miLab™ CER was introduced as a computer-assisted cytology system that uses an AI-based algorithm to present cytologic results on a slide-by-slide basis according to the Bethesda system classification.
- * Screening and treatment of precancerous lesions for secondary prevention of cervical cancer: technology landscape (2024)
- Download
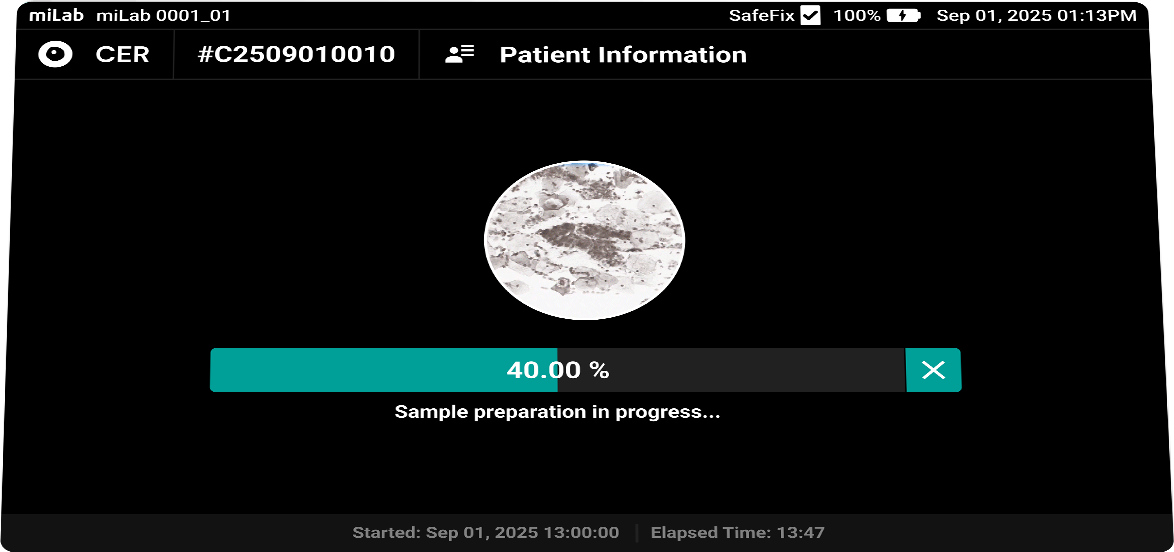
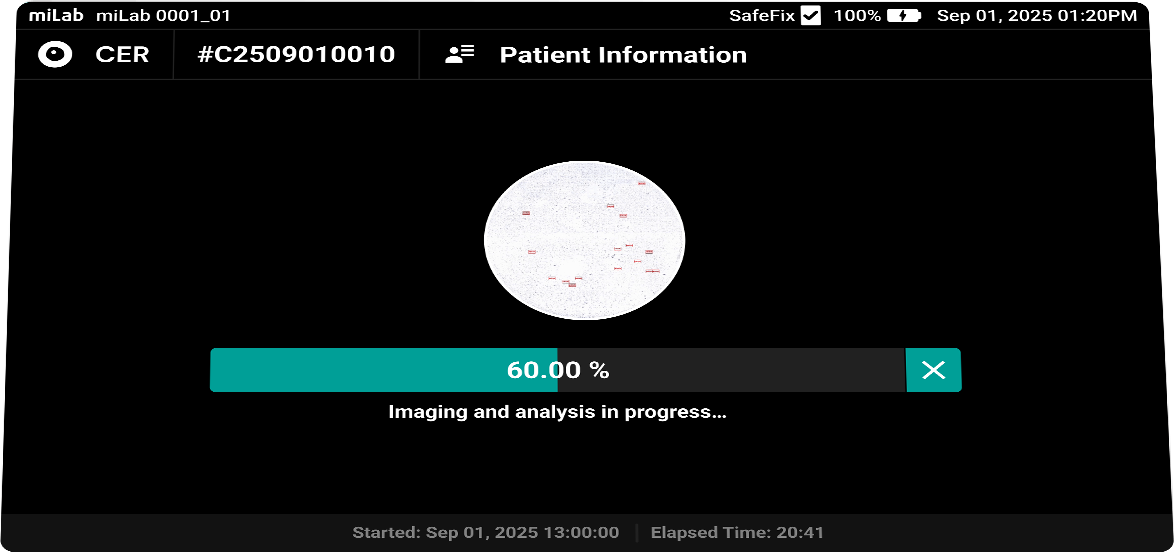
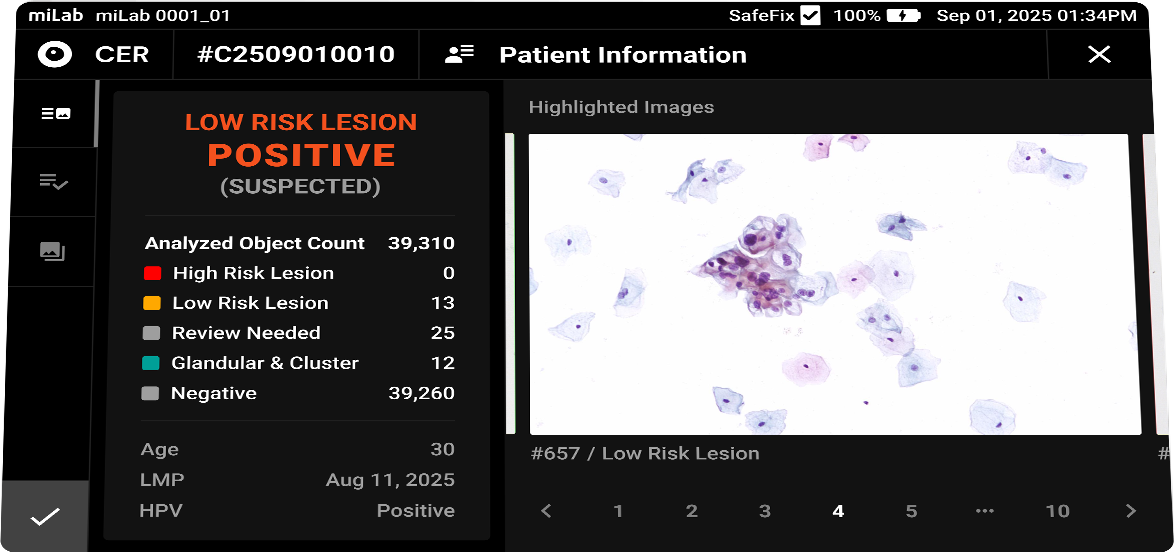
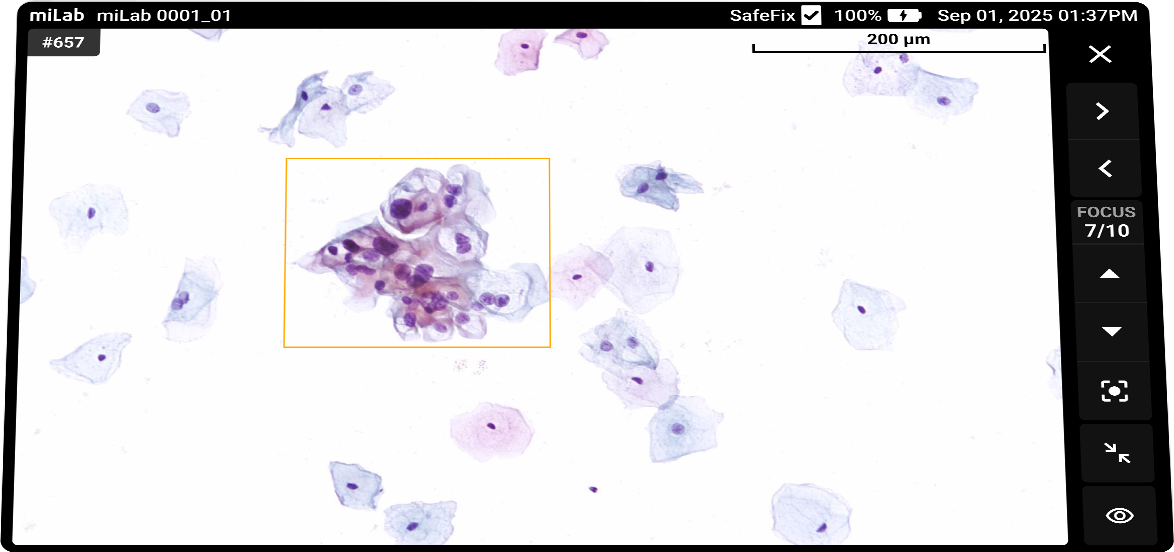
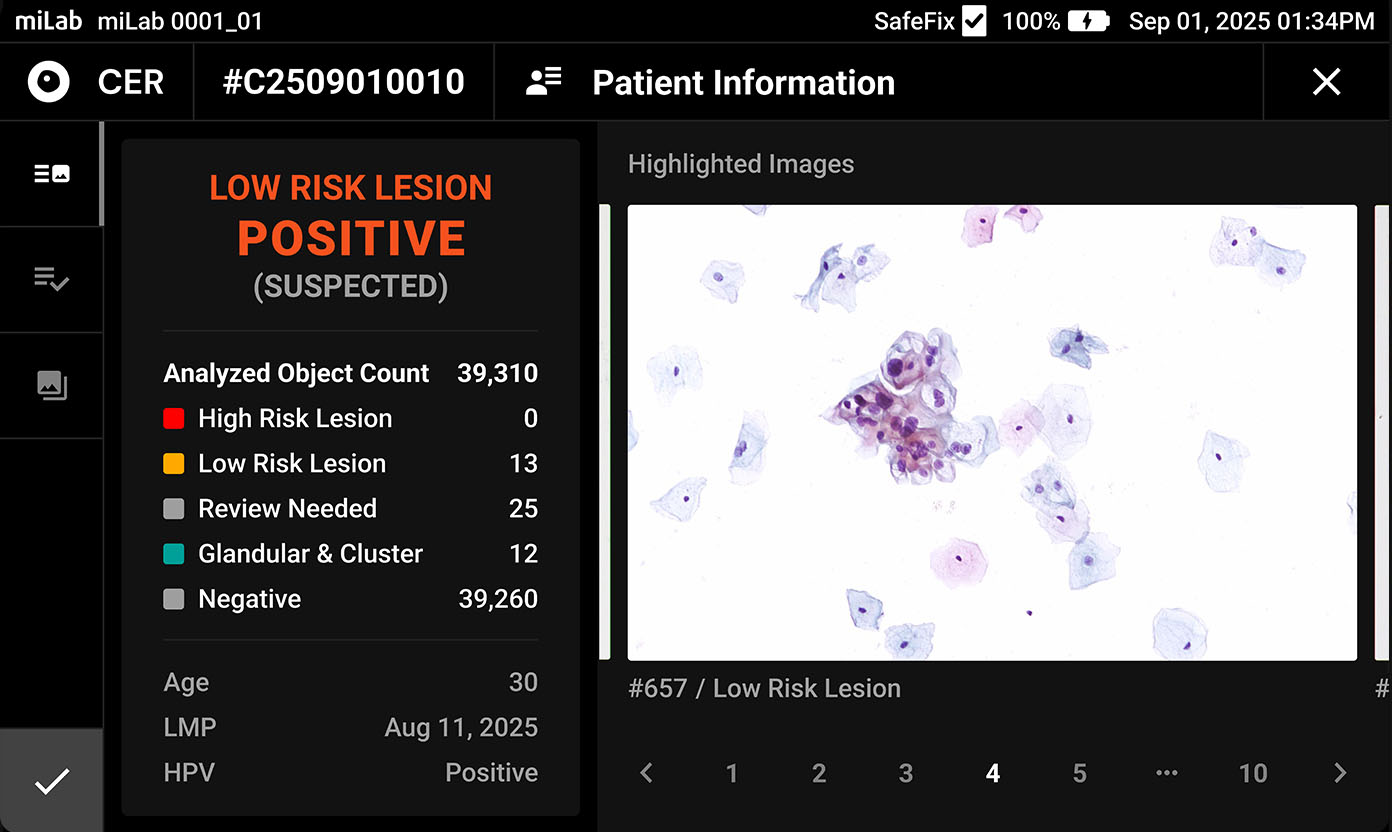
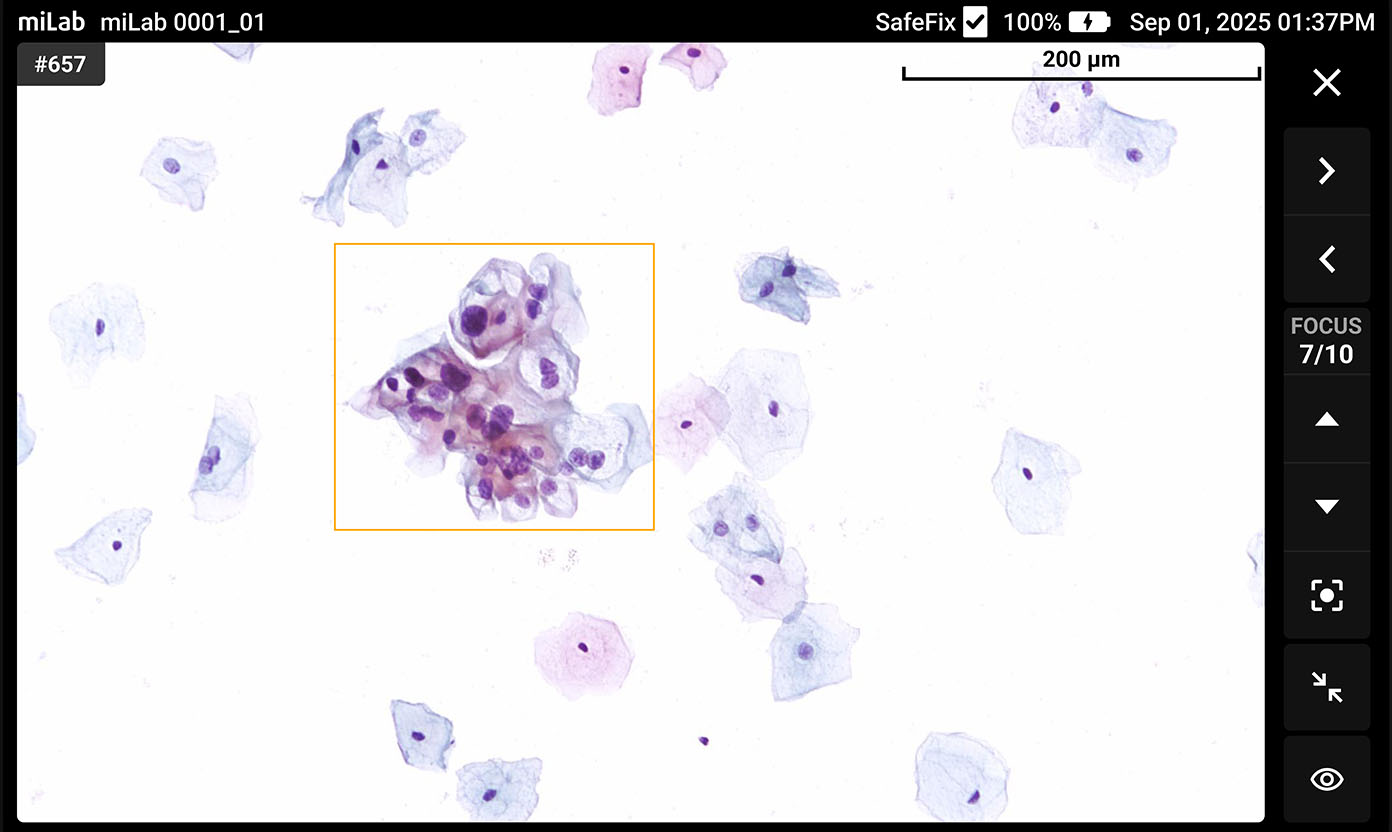
The digital cervical cell analysis solution, miLab™ CER, automates the staining, imaging, and AI-based analysis of smeared cervical cell slides, replacing the traditionally complex workflow and required infrastructure with a single device. The AI algorithm analyzes approximately 25,000 cells per sample, classifies them according to the Bethesda system, and detects signs of pre-cancerous and cancerous lesions. Medical professionals can review and reclassify the results either on-site or remotely via the viewer, enabling them to quickly and accurately determine whether immediate treatment or referral to a higher-level facility is needed.
miLab™ CER focuses on improving access to healthcare for women in settings where early detection of cervical cancer is difficult.
Cervical cancer is the fourth most common cancer among women worldwide, and it is a preventable disease through early screening. However, many women live in environments where early screening is difficult due to a lack of diagnostic infrastructure and trained professionals. The WHO has set a goal to reduce the incidence of cervical cancer by 42% by 2045 compared to 2020 levels, and has called for active participation and technical support from the international community.
Current Status of Cervical Cancer Worldwide
* Year 2021
Pap smear tests and HPV tests are typical cervical cancer diagnostic methods. Despite their advantages, these traditional diagnostic tests have many limitations in terms of infrastructure, operational costs, and reliability of results.
Current Technical Limitations of Cervical Cancer Diagnosis
- In low and middle-income countries, there need to be more experts who can accurately read the results.
- African countries have an average pathologist-to-population ratio of less than 1 per every million, 1/50 of the United States.
- Even in high-income countries such as Canada, the shortage of experts is increasing.
- Papanicolaou staining for cervical cytology consists of 7 types of reagents and 20 detailed work steps, so the proficiency of an expert is essential.
- The possibility of human error depends on the test environment and experts.
- Time required for diagnostic test: At least 2 - 60+ days.
- High workflow costs due to experts, infrastructure,equipment, etc. for accurate diagnosis.
- Difficult to establish a diagnostic laboratory for Pap Smear testing due to resource constraints in low and middle-income countries, which account for 90% of cervical cancer cases.
- Problems with excessive treatment due to HPV tests that cannot quantify the stage of cervical cancer.
End-to-End Automation: Integrating Standard Staining,
Digital Imaging Acquisition, and AI-Powered Analysis.
- Innovative solid staining technology enables standard Pap Smear methods automation
- Replace diagnostic laboratory functionality in one device without additional infrastructure investment.
- Provide Pre-classification of precancerous/cancerous cells based on AI analysis.
- Dramatic improvements of existing workflows
- Dramatically simplifies and automates the Papanicolaou staining process from 20 to 5 steps.
- Fully automated imaging and sample prep processes including staining, cleaning, drying, etc.
- Supports same-day diagnosis and remote diagnostic service
- Test results can be checked in an hour directly at the collection site, allowing for rapid clinical response according to the test results.
- Store digital images as records and support remote diagnosis services.
- Provides reliable early diagnostic tests in low and middle-income countries
- Implement reliable Pap Smear test in resource-constrained environments.
- Prevent over-treatment by providing a clear basis for determining lesions in the field based on the morphological characteristics of cervical cells.
miLab™ CER Enables Medical Institutions with Sample Collection Capabilities to Efficiently and Accurately Perform Cervical Cancer Screening In-house.
- 1. Medical Institutions That Want to Perform Pap Smear Tests In-house
- Target customer
- Medical institutions that are capable of collecting cervical cells, but are not capable of performing Pap smear tests in-house in a country where the primary cervical cancer screening method is a Pap smear.
- Expected Value
- Expand revenue generation opportunities with additional Pap smear tests.
- Background
- Many countries, including the Republic of Korea, operate cervical cancer screening as a state-funded program, and many medical institutions in each country participate in cervical cancer screening programs. However, only a few medical institutions have diagnostic laboratories. Most medical institutions conduct tests by collecting cervical cells and entrusting them to a nearby professional diagnostic laboratory.
- Problem
- Medical institutions unable to perform in-house Pap smear tests must ship samples to an external diagnostic laboratory and pay for the tests. Depending on the examination schedule of the external diagnostic laboratory, it usually takes about 7 to 60 days or more to receive the results.
- Use-Case
- The introduction of miLab™ CER enables those medical institutions capable of collecting cervical cells to perform cervical cancer screening tests with the addition of simple work steps. Since miLab™ CER is a solution that automates the process of sample staining, digital imaging, and AI analysis, medical institutions just need to add the process of smearing and fixing the collected cervical cells on the slide.
- Benefits
- With the introduction of miLab™ CER, institutions can provide accelerated turnaround times, delivering screening results to patients much faster than traditional methods. They can expand their revenue generation opportunities by providing additional tests that they could not previously perform.
- 2. Medical Institutions That Want to Improve the Productivity of Pap Smear Tests
- Target customer
- Medical institutions performing Pap smear tests in a labor-intensive manner.
- Expected Value
- Improve workflow and productivity with the automation of Pap smear tests.
- Background
- A typical Pap smear test is usually a labor-intensive procedure. The sample preparation consists of manual blood smear, fixation, staining, washing, and drying, and then experts check the shape and distribution of cervical cells one by one through a microscope. Papanicolaou staining is, in particular, quite complicated with 20 steps of tasks.
- Problem
- Due to the labor-intensive method, the greater the number of Pap smear tests, the higher the level of physical fatigue felt by experts, and the higher the possibility of human error. It is difficult to perform more tests maintaining employee and time constraints because each medical institution has fixed working standards for Pap smear tests.
- Use-Case
- Medical institutions looking to improve the productivity of Pap smear tests can use miLab™ CER to automate some parts of experts’ tasks. miLab™ CER automates the most complicated and error-prone Papanicolaou dyeing, digital imaging, and AI analysis processes. Experts can focus on reviewing and finalizing the AI-analyzed results on the monitor screen without taking care of complicated staining or locating cervical cells one by one. While experts are away, other staff members can work with miLab™ CER. When experts come back to work, they can review and finalize the pre-analyzed results stored in the database.
- Benefits
- miLab™ CER maximizes operational efficiency, allowing institutions to significantly increase test volumes without additional staffing or extended hours. Medical institutions can respond to the growing demand for tests by introducing miLab™ CER without hiring more experts. Higher productivity is more valuable in high-income countries where labor costs are high.
- 3. Medical Institutions That Want to Improve the Accuracy of Pap Smear Tests
- Target customer
- Medical institutions that have their own diagnostic laboratories but conduct Pap smear tests with low-skilled experts.
- Expected Value
- Enhancing quality assurance and quality management systems to improve the reliability of Pap smear tests.
- Background
- Typically, experts performing Pap smear testing require specialized knowledge and experience. In addition to completing professional education above the university and passing the national qualification exam, practical education and training are also required. In countries with well-equipped medical education facilities and systems such as the Republic of Korea, the level of experts is maintained. However, in low and middle-income countries, medical education facilities and systems are insufficient.
- Problem
- Because the Pap smear test is carried out in a labor-intensive manner, the analysis and judgment results vary depending on the proficiency of the expert. Incorrect analysis results from the Pap smear test lead to incorrect diagnosis and treatment of the patient. Healthcare organizations can compare the results by having multiple experts perform tests simultaneously to ensure that the results are accurate, or have a proven expert verify the results. However, all of these methods incur additional cost burdens, such as hiring highly qualified personnel.
- Use-Case
- For healthcare organizations that want to improve the accuracy of Pap smear testing, miLab™ CER can be included in their quality control system. miLab™ CER standardized Pap smear's core processes from Papanicolaou staining to AI analysis. Therefore, the reliability of experts can be confirmed by comparing the results of the Pap smear test with the results of the miLab™ CER. In addition, digital data generated by miLab™ CER can be used for education and training materials for low-skilled experts.
- Benefits
- For healthcare organizations that want to improve the accuracy of Pap smear testing, miLab™ CER can be included in their quality control system. miLab™ CER standardized Pap smear's core processes from Papanicolaou staining to AI analysis. Therefore, the reliability of experts can be confirmed by comparing the results of the Pap smear test with the results of the miLab™ CER. In addition, digital data generated by miLab™ CER can be used for education and training materials for low-skilled experts.
- 4. Medical Institutions That Want to Perform the Remote Pap Smear Tests
- Target customer
- Medical institutions that are capable of collecting cervical cells without their own diagnostic laboratory, however, don't have higher-level hospitals with experts nearby to ask for consignment examinations.
- Expected Value
- Improving medical access for patients marginalized from the existing medical system.
- Background
- In remote and mountainous regions where physical access is not easy or in low and middle-income countries where specialized educational facilities are scarce, there are not enough experts to conduct Pap smear tests. In some areas, there are no medical institutions capable of performing the Pap smear tests.
- Problem
- Residents living in such areas, where it is difficult to take the Pap smear test, have the inconvenience of traveling a long distance to get a cervical cancer screening test. Due to this inconvenience, they may not receive regular cervical cancer screening tests, which leads to being exposed to the risk of disease.
- Use-Case
- Medical institutions capable of collecting cervical cells in such areas can share digital data to ask remote experts for precise readings.
- Benefits
- With the introduction of miLab™ CER, those medical institutions can improve residents' access to cervical cancer screening to help residents diagnose cervical cancer early and receive appropriate and timely treatment.
- 5. The Government That Wants to Switch to a More Reliable Cervical Cancer Screening System
- Target customer
- The government of low and middle-income countries where the number of cervical cancer cases and deaths are serious but rely on VIA tests as cervical cancer screening methods due to a lack of national diagnostic laboratory infrastructure and experts.
- Expected Value
- Establishing a reliable basis for early screening of cervical cancer to reduce the number of cervical cancer cases and deaths in the country while minimizing investment in infrastructure and experts.
- Background
- A Pap smear is the most common and accurate screening method for cervical cancer, but it has limitations in diagnostic laboratory infrastructure and reliance on experts. More than 50 low and middle-income countries worldwide use the VIA method as a screening method for cervical cancer due to lack of resources, or do not specify a screening method at the government level.
- Problem
- The VIA method is non-scientific since it depends on the visual checking of inspectors. The WHO does not recommend this screening method due to low accuracy and concerns over subjective judgment by inspectors. In countries that use the VIA method for cervical cancer screening, it is difficult to accurately determine the patient's condition, making it difficult to achieve early diagnosis and early treatment of cervical cancer.
- Use-Case
- Governments that rely on VIA methods or do not specify a method for cervical cancer screening can use miLab™ CER to designate the Pap smear tests as the national standard for cervical cancer screening. miLab™ CER minimizes medical institutions' investment in diagnostic laboratory infrastructure to perform Pap Smear tests, and can establish a cervical cancer screening system that can best utilize a small number of experts by improving workflows and providing remote diagnostic services. By providing a wealth of materials for educating and training experts based on accumulated digital data, introducing miLab™ CER can contribute to fostering experts in the country.
- Benefits
- With the introduction of miLab™ CER, these countries will be able to shift their cervical cancer screening methods to the most common and reliable Pap Smear tests with minimal diagnostic laboratory infrastructure and expert investment. This allows people to accurately diagnose cervical cancer early and establish a public health system that can receive appropriate treatment.
- 6. Other Cases
- miLab™ CER is a widely applicable solution for various environments and situations. Do not hesitate to contact us anytime if you are interested in using miLab™ CER products. With Noul you can create a healthcare environment supporting women in early and regular cervical cancer screening; improving health and saving lives.
Stain, Scan, and
Classify cells in 20 mins
- miLab™ CER Specification
- Sample Preparation
Sample type Cervicovaginal sample smeared with LBC (Liquid-Based Cytology) Staining type Gold-standard Papanicolaou staining
- Imaging
Lens 20x magnification Focus Provides 10 z-stack images with the best focus for each cell Imaging Automatically detects an average of 25,000 cells across the entire LBC smear field
- Analysis
Classification NILM, ASCUS, LSIL, ASC-H, HSIL, SCC Images Field-binding images classified based on the presence or absence of atypical cells
- Total Test Time
up to 30 minutes
- Remote Review
Viewing Functions AI classification, Field images, Reclassification and Report
- Product Code
Product Code miLab™ Platform DMLA miLab™ Cartridge CER CCEA miLab Viewer™ SMVA SafeFix™ CER CSCA miLab™ Slide Case ASCA